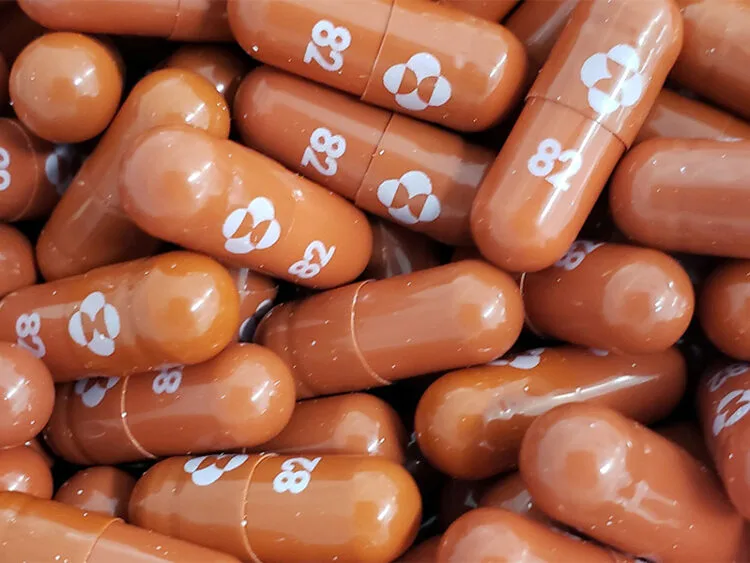
The government is in talks of procuring a COVID-19 antiviral pill developed by US drug-maker Merck & Co called Molnupiravir. In a recent trial, the data showed that the treatment halves the risk of hospitalisation or death for those who contracted the virus and took the pill twice a day for five days.
Molnupiravir is designed to introduce errors into the genetic code of the virus and would be the first of its kind. However, while Merck’s is currently the closest to achieve approval, there are numerous pharmaceutical companies racing to develop similar pills, including famed vaccine-producer Pfizer.

According to Merck, the antiviral pill is effective against all variants of COVID-19, including the devastating Delta variant that has a higher transmissibility. “This is going to change the dialogue around how to manage COVID-19,” said Merck CEO Robert Davis.
Health Minister Khairy Jamaluddin announced the potential purchase on Twitter and hints that there may be other experimental treatments being looked at. The move is part of Malaysia’s preparations to transition into an endemic phase, what the minister calls, “living with COVID.”

Last month, Khairy said the government is no longer using the conventional herd immunity formula of 80% as the Delta variant had “thrown out the herd immunity calculation.” The adult population has reached an 87% rate of full vaccination as of today, with the country sprinting towards allowing interstate travel when that figure reaches 90%.
(Sources: KJ/Twitter, Reuters, AstroAwani // Images: Merck & Co, MOSTI)