UPDATE (5 MAR / 4:20 PM): Health Minister Khairy Jamaluddin has confirmed that Malaysia will receive 110,000 patient-courses of Paxlovid in two weeks. As for the treatments from Merck and AstraZeneca, the government also has plans to buy them but it has not been finalised yet.
ORIGINAL STORY (4 MAR / 1:25 PM):
Director-General of Health Dr Noor Hisham Abdullah has announced that the Drug Control Authority (DCA) has given conditional approval for the Paxlovid COVID-19 treatment drug made by pharmaceutical firm Pfizer. The product registration holder is Pfizer Malaysia and it is only approved for use on adults over 18 with a high risk of contracting severe symptoms from the virus.
Paxlovid is a combination treatment using nirmatrelvir and ritonavir, taken orally and has been shown in clinical trials to be highly effective at preventing hospitalisation and death. Dr Noor Hisham reminded the public that the treatment is not a replacement for the vaccine and people should still do their best to follow SOPs.
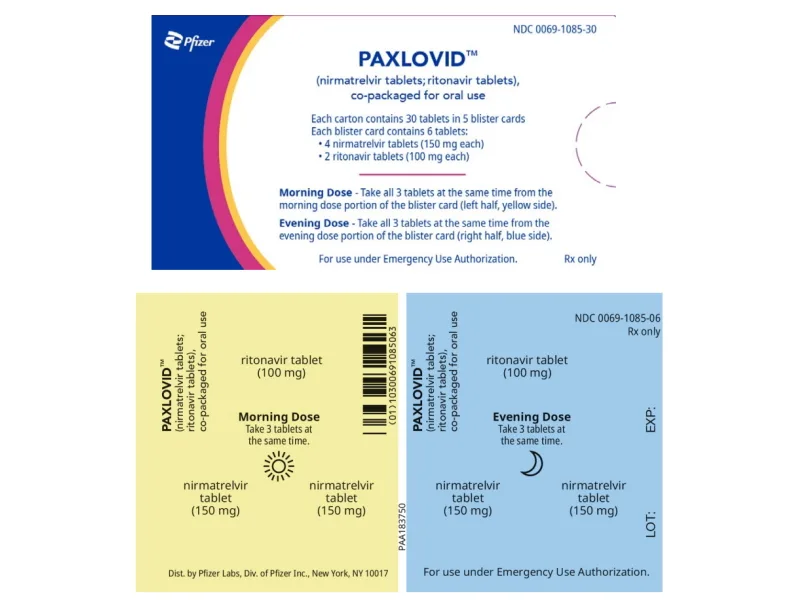
The Ministry of Health (MoH) had already announced back in December that it was planning to procure the drug for use here, but did not reveal how many courses will be purchased. Aside from Paxlovid, Malaysia also plans to buy treatment drugs from AstraZeneca and Merck, although the latter will only submit its pill for approval sometime this year. Another COVID-19 treatment called Ronapreve is also already in use in Sarawak.
The Pfizer treatment has already been approved in the US, UK, the European Union, Israel, South Korea, Canada, China, and Singapore. The price of the drugs have not been revealed, but for reference, it cost the US government US$530 (~RM2200) for each five-day course which is cheaper than Merck’s molnupiravir at US$700 (~RM2900) for a five-day course.
(Source: Director-General of Health.)